Jeffrey A. Niezgoda, MD, FACHM, MAPWCA, CHWS; Thomas E. Serena, MD, FACS, FACHM, MAPWCA; and Marissa J. Carter, PhD, MAPWCA |
BACKGROUND: The late effects of radiation therapy following the treatment of cancer is a well-known consequence. Evidence increasingly supports the use of hyperbaric oxygen (HBO) as an adjunctive treatment in a variety of radiation injuries. OBJECTIVE: To present the findings of a new registry of radiation injuries that was developed to evaluate the outcomes and treatment parameters of HBO treatment (HBOT) when applied to patients experiencing the late effects of radiation therapy. DESIGN: Observational cohort. SETTING: Hyperbaric oxygen clinical treatment facilities in the United States. PATIENTS: A total of 2538 patients with radiation-induced injuries. MEASUREMENTS: Injury type, patient age, gender, diabetes, end-stage renal disease, collagen vascular disease, coronary artery disease/peripheral vascular disease, on anticoagulant medication, on systemic steroid medication, the patient is current smoker, patient abuses alcohol, symptoms reported, duration of symptoms, symptom progression prior to HBOT, transfusion units, HBOT time, HBOT count, HBO chamber pressure, HBO time in the chamber, and patient outcomes. | RESULTS: A total of 2538 patient entries with 10 types of radiation injuries were analyzed. The 5 most common injuries were osteoradionecrosis (33.4%), dermal soft tissue radionecrosis (27.5%), radiation cystitis (18.6%), radiation proctitis (9.2%), and laryngeal radionecrosis (4.8%). Clinical outcomes following HBOT were positive with symptoms that improved or resolved varying from 76.7% to 92.6%, depending on injury type. Overall, although the mean symptom improvement score between some groups is statistically significant, the differences are probably not clinically meaningful. Patients with osteoradionecrosis had the highest mean symptom improvement score (3.24) compared with a mean of 3.04 for laryngeal radionecrosis. LIMITATIONS: Limited data were available on patient comorbidities and symptom severity. CONCLUSIONS: Outcomes from a large patient registry of radiation-induced injuries support the continued therapeutic use of HBOT for radiation injuries. KEYWORDS: radiation injury, hyperbaric oxygen therapy, late effects of radiation, osteoradionecrosis, American College of Hyperbaric Medicine |
ADV SKIN WOUND CARE 2016;29:12-9
Radiation injuries are an unfortunate consequence that can occur months or years following cancer treatment, resulting in complications due to tissue ischemia.1-3 Osteoradionecrosis (ORN), laryngeal radionecrosis (LRN), dermal soft tissue radionecrosis (STRN), radiation-induced cystitis, and proctitis are among the most common radiation injuries.1,4 Mucosal tissues (such as gastrointestinal and urinary) are at high risk of such injuries because of their fragility and rapid cellular turnover.
Hypoxia in the bladder leads to cystitis with pain, hematuria, excessive urination, and incontinence, with severe complications, including ulceration and fibrosis.5,6 Proctitis is similar to cystitis but occurs in the rectal tissues, the most distressing symptoms of which include pain, fistulae, ulceration, and rectal bleeding.7-9 Radiation-induced tissue changes commonly occur in cancer patients, with as many as 10% developing ORN, STRN, or cystitis,1,5,6, and up to 20% of those receiving radiation to the pelvic area developing proctitis.7,8
Conservative treatment varies depending on the type of injury. For ORN, irrigation with an antiseptic, good oral hygiene, and debridement are often indicated.10 Analgesics can be administered for pain and antibiotics if the infection is present. In advanced cases, surgical treatment is necessary, including reconstructive surgery and skin grafting.10,11 For LRN, nonsurgical and surgical options aim for laryngeal salvage.12 For less severe cystitis, exercises, muscle relaxants, and anticholinergic drugs are indicated.5,6 In more severe cases, bladder irrigation, intravesical instillation, oral or intravenous agents, electrocoagulation or laser cauterization, iliac artery embolization, urinary diversion, and cystectomy are options, although the consequences of surgery can be catastrophic.5,6 Conventional treatment for proctitis is similar to that of cystitis, and surgery is also used as a last resort because of the potential for significant complications.13 Anti-inflammatory medications, formalin, steroids, and sucralfate may be effective for severe proctitis, whereas laser treatment and argon plasma thermal coagulation are commonly applied to control rectal bleeding.13,14
Evidence, including 2 systematic reviews,1,15 supports the use of hyperbaric oxygen (HBO) as an adjunctive treatment for radiation injuries, with up to 80% of patients benefitting.1,16 In fact, HBO stimulates angiogenesis and osteogenesis and reduces the inflammatory fibrosis that is present with radiation injuries.17 In addition, HBO can also prevent and treat ORN1,18 and is supported by 2 randomized controlled trials (RCTs).19,20 A large review of prospective outcomes in 411 patients found that 94% of patients with ORN benefitted from HBO, as well as 76% of patients with dermal STRN, 82% with LRN, and 89% with cystitis.21 There are also 7 case series that support the use of HBO in the radiation-affected larynx.22 A systematic review also supports the use of HBO in cystitis as the most effective treatment, along with intravesical hyaluronic acid.23 Hyperbaric oxygen therapy reduces bladder inflammation and increases tissue vascular density affected by cystitis.1 A small, randomized phase 2 trial16 and 1 RCT24 also support the use of HBO for radiation-induced proctitis.12,25
The objective of this current study is to evaluate the outcomes and treatment parameters of HBO treatment (HBOT) when applied to patients with radionecrosis injury included in a radionecrosis registry.
As early as 2004, the American College of Hyperbaric Medicine (ACHM) began to receive numerous notifications from its membership that several national insurance providers had determined that HBOT for radiation necrosis would be categorized as “Bexperimental-investigational,” subsequently resulting in submitted claims denials. The results of these determinations had they been left unchallenged would have eliminated the use of HBOT for patients with radiation injuries. Informal surveys have shown that at most hyperbaric centers in the United States, nearly one-half of patients receiving HBO are being treated for radiation injury (Undersea and Hyperbaric Medical Society, Delayed Radiation Injury; available at https://www.uhms.org/11-delayedradiation-injury-soft-tissue-and-bony-necrosis.html). Thus, the reimbursement determinations would have denied the benefits of HBO to patients who were experiencing the late effects of radiation injury. The decisions would also threaten the clinical practice of hyperbaric medicine.
In 2007, the ACHM launched the Radionecrosis Research Registry (RRR). The goal of this registry project was to collect outcome data on patients with radionecrosis injury who were treated with HBO in an observational cohort study and to report clinical outcomes data in the form of a patient-centered registry. The hope was that historical observations of significant clinical improvement noted in patients with radionecrosis treated with HBOT would be documented and confirmed with an analysis of multicenter clinical outcome data. The registry findings would serve to address the prior concerns expressed by payors and become an indexing platform for monitoring patient-centered outcomes now and in the future.
Literature was searched on PubMed using the search terms “radionecrosis,” “radiation injuries”, “Bproctitis”, “Bcystitis”, “treatments”, and “hyperbaric oxygen” for the time frame of January 1, 1985, through May 31, 2014. The search was narrowed down to cohort studies and RCTs. Evidence was limited to 4 RCTs, including 3 RCTs on the effect of HBOT on ORN,19,20,26 1 RCT on the effect of HBOT on proctitis,24 and 1 cohort study on ORN reconstructive surgery.11 Outcomes reported in these studies served as the basis for the design of the registry and the references provided herein.
The registry was created in PHP: Hypertext Preprocessor (The PHP Group), backed by a MySQL database (Oracle Corporation, Redwood Shores, California). All communication to and from the site was encrypted via 128-bit Advanced Encryption Standard (National Institute of Standards and Technology, Gaithersburg, Maryland). On the home page, a site visitor registered as a new institution. After site approval, the medical director was set up as the primary user for the institution, and additional data-entry users could be created using the institution’s identification. Once logged in, a user entered a new case record. The process for entering a new case included the collection of deidentified patient information, followed by symptom and treatment data for the selected radiation injury. The user reviewed all the data entered for the case prior to final submission and storage in the database. Once data entry was finalized, no updates were allowed.
At the time of analysis, the registry contained entries for 2577 patients that presented for HBOT between 2004 and 2009. Data were provided by 314 users at 108 HBO clinical treatment facilities (clinics, hospitals, and medical centers). All entries were evaluated for data integrity to determine eligibility for study inclusion.
Based on a collaborative effort with a participating clinical site, the study was approved by the Aurora Health Care Institutional Review Board ([IRB] Milwaukee, Wisconsin) as an exempt protocol (45CFR46.101) and granted IRB waiver of Health Insurance Portability and Accountability Act authorization (45CFR 164.512). The study required informed consent acknowledging the use of deidentified data for analysis and publication.
Under symptom time (years), 2 data entries were related to the actual year that the symptoms started (eg, 2009) and not duration, and these values were deleted. Two other symptom entries were larger than the patient’s age, and these values were also deleted, as it was assumed that these figures were incorrect. There were also 12 entries with symptoms of more than 10 years_ duration that was kept; in 1 instance, the problem would have started at a patient age of 9 years.
Thirty-nine patients had a patient age of 1 year; it was assumed that these were incorrect entries, as symptom durations were sometimes longer than 1 year, and some patient comorbidities/ characteristics, such as coronary artery disease (CAD) or peripheral vascular disease (PVD), type 2 diabetes, or smoking, would be very unlikely. These cases were deleted, leaving 2538 patients in the database to be analyzed from the original count of 2577.
The following variables were extracted from the registry: type of injury, patient age (years), gender, diabetes (type 1 or 2 or no diabetes), end-stage renal disease (ESRD), collagen vascular disease, CAD or PVD, anticoagulant medication, on systemic steroid medication, the patient is current smoker, patient abuses alcohol, symptoms reported, duration of symptoms (months), symptom progression prior to HBOT (stable or progressing), transfusion units (count at a patient level), HBOT time (months of treatment), HBOT count (number of sessions), HBO chamber pressure (atmospheres absolute), HBO time in the chamber (minutes, each session), and patient outcomes (worse, no change, improved, resolved).
Continuous variables were reported as means (SD) if data were normal or median and range (and interquartile range, if relevant) if data were severely non-normal as demonstrated by the Wilk-Shapiro test. Some exceptions were made for the Wilk-Shapiro test violation as the test tends to be overly sensitive for large samples, and inspection of histograms can provide a better assessment. Categorical variables were reported as counts and proportions (percentages).
Because not all patients reported symptoms, and many patients had multiple symptoms, the possible outcomes (worse, no change, improved, and resolved) were translated into a scale of 1 to 4, and the mean symptom improvement score was calculated for each patient. The same scale was then used to interpret the mean score for each patient.
Comparisons between injury types were conducted using Kruskal-Wallis tests for nonnormal continuous variables (1-way analysis of variance [ANOVA] by ranks is a nonparametric method for testing whether samples originate from the same distribution), ANOVA for normal continuous variables, and W2 test for nominal categorical variables. For testing related variables, a full Bonferroni correction (a method used to counteract the problem of multiple comparisons) was applied to control the familywise error rate.
All statistical analysis was conducted using SPSS Statistics 21 (IBM, Chicago, Illinois). An a of .05 was considered statistically significant. All tests were 2-sided.
This study was funded by the ACHM and the Eric P. Kindwall, MD, Foundation; the ACHM had no role in the actual analysis.
Of the 2538 patients analyzed in the registry, 3 had missing data. There were 10 types of radiation injuries registered. The 5 most common radiation injuries were analyzed: ORN (33.4%), dermal STRN (27.5%), cystitis (18.6%), proctitis (9.2%), and LRN (4.8%) (Table 1).
Patient demographics, characteristics, and comorbidities are presented in Table 2. The difference in patient age by type of injury was significant (P G .001), as was the difference in the ratio of males to females by type of injury (P G .001). There were significant differences among the injuries between type 1 and 2 diabetes (%) and the percentage of patients with diabetes (P G .001). The prevalence of ESRD was low, with no meaningful correlation found between having ESRD and diabetes. There were significant differences between patients with different types of injuries regarding the prevalence of CAD or PVD (P G .001), with the highest prevalence in patients with cystitis (27.6%) and the lowest in patients with ORN (15.5%). There was significant variation in the percentage of patients on anticoagulants by type of injury, with patients with cystitis being the highest (19.5%) and patients with ORN being the lowest (10.6%; P G .001). The percentage of patients on steroids varied by as much as 2.5-fold, with patients with cystitis the lowest (2.6%) and patients with dermal STRN the highest (6.4%), but was
Table- 1: Types of Injury
Type of Injury | n | % |
Osteoradionecrosis | 847 | 33.4 |
Dermal soft tissue radionecrosis | 698 | 27.5 |
Cystitis | 473 | 18.6 |
Proctitis | 232 | 9.2 |
Laryngeal radionecrosis | 121 | 4.8 |
Cerebral damage | 49 | 1.9 |
Vaginosis | 43 | 1.7 |
Colitis | 35 | 1.4 |
Esophagitis | 22 | 0.9 |
Enteritis | 15 | 0.6 |
Subtotal | 2535 | 99.9 |
Missing data | 3 | 0.1 |
Total | 2538 | 100.0 |
Table- 2: PATIENT DEMOGRAPHICS, CHARACTERISTICS, AND COMORBIDITIES BY TYPE OF INJURY a
Patient Characteristics | Osteoradionecrosis | Dermal Soft Tissue Radionecrosis | Cystitis | Proctitis | Laryngeal Radionecrosis |
Mean age (SD), y | – | – | – | – | 61.1 (10.5) |
Median age (range), y | 61 (8Y88) | 64 (7Y88) | 73 (23Y88) | 72 (30Y88) | – |
Gender, % | |||||
Male | 68.2 | 47.2 | 84.4 | 81.4 | 73.6 |
Female | 31.8 | 52.8 | 15.6 | 18.6 | 26.4 |
Diabetes, n (%) | |||||
Type 1 | 26 (3.1) | 44 (6.3) | 41 (8.7) | 14 (6.1) | 7 (5.8) |
Type 2 | 68 (8.0) | 70 (10.0) | 63 (13.3) | 28 (12.2) | 13 (10.7) |
Tota | 94 (11.1) | 114 (16.3) | 104 (22.0) | 42 (18.1) | 20 (16.5) |
End-stage renal disease, n (%) | 5 (0.6) | 8 (1.3) | 6 (1.4) | 3 (1.4) | 1 (0.8) |
Collagen vascular disease, n (%) | 16 (2.2) | 19 (3.1) | 9 (2.1) | 6 (3.0) | 1 (0.9) |
CAD/PVD, n (%) | 116 (15.5) | 139 (21.8) | 119 (27.6) | 48 (23.1) | 27 (23.4) |
On anticoagulants, n (%) | 79 (10.6) | 85 (13.4) | 86 (19.5) | 39 (18.1) | 16 (13.9) |
On steroids, n (%) | 23 (3.1) | 40 (6.4) | 11 (2.6) | 11 (5.4) | 5 (4.3) |
Smoker, n (%) | 186 (23.5) | 93 (14.6) | 50 (11.4) | 26 (12.3) | 25 (21.4) |
Alcohol abuse, n (%) | 85 (11.0) | 32 (5.1) | 13 (3.0) | 7 (3.4) | 6 (5.2) |
Abbreviations: CAD, coronary artery disease; PVD, peripheral vascular disease. a Where patient age is nonnormal, medians and ranges are provided.
not significant after adjustment for multiplicity. Patients with ORN and LRN smoked nearly twice as much as other patients (P G .001). Patients with ORN also had a 2 to 3 times higher rate of alcohol abuse compared with other groups (P < .001).
There were 16 symptoms of radiation injuries reported by patients with the 5 major radiation injuries: ORN, STRN, cystitis, proctitis, and LRN. These pre-HBOT symptoms are presented by percentage in Table 3. Some symptoms were very specific to the type of injury; for example, diarrhea and blood in stool were reported by all patients with proctitis, hematuria in all patients with cystitis, and exposed alveolar bone in all patients with ORN. All patients with cystitis and proctitis reported repeated cystoscopy or endoscopy procedures because of their radiation injury prior to HBOT. Nearly all patients experienced pain and dermal injury manifesting as fistula or wound healing compromise (ORN, STRN, cystitis, and LRN, 100%; proctitis, 98.2%).
There was no significant difference in symptom duration among injuries, which had a mean duration of 75 (SD, 1221.1) months (median, 6 months; interquartile range [IQR], 10 months). Symptom progression for each type of injury prior to HBOT was highest for cystitis (85.8%) and lowest for ORN (63.4%) (Table 4). Differences between groups were significant (P > .001).
Eight percent of patients with cystitis and 10% with proctitis required transfusion; the respective medians and ranges were 2 (1-10 units) and 3 (1-14 units).
The mean HBOT treatment time for ORN was 2.2 (SD, 1.57) months, for dermal STRD was 2.1 (SD, 1.87) months, for cystitis was 2.1 (SD, 1.45) months, for proctitis was 2.3 (SD, 1.16) months, and for LRN was 1.8 (SD, 0.91) months. Although the distributions failed the Wilk-Shapiro test, inspections of the histograms suggested that the distributions were normal enough that they would be suitable for ANOVA. The Levene test for the equality of group variances was not significant, which means that the F statistic could be used to test overall mean differences between groups. However, the test was not significant.
The median count of HBO sessions for ORN was 30 treatments (range, 1-80; IQR, 8), for dermal STRN was 30 (range, 1-90; IQR, 18), for cystitis was 30 (range, 1-168; IQR, 14), for proctitis was 40 (range, 2-100; IQR, 20), and for LRN was 30 (range, 1-60; IQR,
Table- 3: SYMPTOMS BY PERCENTAGE FOR THE 5 MAJOR RADIATION INJURIES FOR THOSE PATIENTS REPORTING SYMPTOMS
Symptom | ORN | STRN | Cystitis | Proctitis | LRN |
Hematuria | 0 | 0 | 100 | 0 | 0 |
Pain/dysphagia | 100 | 100 | 100 | 98.2 | 100 |
Frequency | 0 | 0 | 100 | 0 | 0 |
Fistula/wound | 100 | 100 | 100 | 98.2 | 100 |
healing compromise | |||||
Repeat cystoscopy/ | 0 | 0 | 100 | 100 | 0 |
endoscopy | |||||
Diarrhea | 0 | 0 | 0 | 100 | 0 |
Tarry/bloody stool | 0 | 0 | 0 | 100 | 0 |
Bleeding | 0 | 0 | 0 | 1.7 | 0 |
Drainage | 100 | 100 | 0 | 0 | 0 |
Exposed alveolar bone | 100 | 0 | 0 | 1.7 | 0 |
Ulceration | 0 | 0 | 0 | 1.7 | 0 |
Inability to swallow | 0 | 0 | 0 | 0 | 100 |
Speech compromise | 0 | 0 | 0 | 0 | 100 |
Abbreviations: LRN, laryngeal radionecrosis; ORN, osteoradionecrosis; STRN, soft tissue radionecrosis.
Table- 4: SYMPTOM PROGRESSION PRIOR TO HYPERBARIC OXYGEN THERAPY
Type of Injury | Stable (%) | Progressing (%) | Total (%) |
Cystitis | 62 (14.2) | 374 (85.8) | 436 (100.0) |
Proctitis | 44 (20.9) | 167 (79.1) | 211 (100.0) |
Osteoradionecrosis | 289 (36.6) | 501 (63.4) | 790 (100.0) |
Dermal soft tissue | 145 (21.5) | 529 (78.5) | 674 (100.0) |
radionecrosis | |||
Laryngea | 20 (17.7) | 93 (82.3) | 113 (100.0) |
radionecrosis | |||
Total | 560 (25.2) | 1664 (74.8) | 2224 (100.0) |
17). The difference in the number of HBO sessions between injury groups was significant (P < .001).
The HBOT chamber pressure for each type of injury is shown in Table 5. There was significant variation between types of injury and chamber pressure, with patients with ORN having higher chamber pressures and patients with laryngeal radionecrosis having lower chamber pressures (P < .001). The median session time for all groups was 90 minutes with very little variation between groups.
Patient outcomes after HBOT are reported in Table 6. Only 4.4% of patients had no reported post-HBO outcomes in regard to any of their symptoms. The percentage of patients who experienced an improvement or resolution of their symptoms did differ by type of radiation injury, with injury types of proctitis and ORN overall having the best results (92.6% and 92.0%, respectively) and LRN having the least benefit (76.7%) (P = .0004). Outcomes and percentage change in major symptoms following HBOT reported by injury types, ORN, STRN, cystitis, and proctitis, are graphically illustrated in Figures 1 to 5.
This observational cohort study of the RRR included 10 different types of radiation injuries from 2538 patients, which is one of the largest reported radiation injury data sets to date. Osteoradio
Table- 5: HYPERBARIC OXYGEN TREATMENT CHAMBER PRESSURE APPLIED FOR EACH TYPE OF INJURY
Hyperbaric Oxygen Pressure (ATA) | ||||
Type of Injury | 2.0 | 2.4 | 2.5 | Total |
Cystitis (%) | 63 (14.0) | 271 (60.1) | 117 (25.9) | 451 (100.0) |
Proctitis (%) | 37 (16.7) | 120 (54.1) | 65 (29.3) | 222 (100.0) |
Osteoradionecrosis (%) | 80 (9.7) | 469 (56.8) | 276 (33.5) | 825 (100.0) |
Dermal soft tissue | 105 (15.7) | 371 (55.5) | 192 (28.7) | 668 (100.0) |
radionecrosis (%) | ||||
Laryngeal radionecrosis (%) | 21 (17.8) | 50 (42.4) | 47 (39.8) | 118 (100.0) |
Total (%) | 306 (13.4) | 1281 (56.1) | 697 (30.5) | 2284 (100.0) |
Abbreviation: ATA, atmospheres absolute
Table- 6: OUTCOMES AFTER HYPERBARIC OXYGEN THERAPY ON A PATIENT BASIS
Type of Injury | Mean Symptom Improvement Score | SD | Improved/ Resolved,a % | Total, n (%) |
Cystitis | 3.24 | 0.675 | 84.5 | 432 (22.2) |
Proctitis | 3.25 | 0.552 | 92.6 | 217 (11.1) |
Osteoradionecrosis | 3.28 | 0.572 | 92.0 | 588 (30.2) |
Dermal soft tissue | 3.16 | 0.649 | 86.7 | 608 (31.2) |
radionecrosis | ||||
Laryngeal | 3.04 | 0.617 | 76.7 | 103 (5.3) |
radionecrosis | ||||
Total | 3.22 | 0.623 | 87.9 | 1948 (100.0) |
The outcome scale is as follows: 1 = worse, 2 = no change, 3 = improved, 4 = resolved. a The percentage of patients whose mean scores for symptoms post-hyperbaric oxygen treatment were between 3 and 4. | ||||
necrosis, dermal STRN, cystitis, proctitis, and laryngeal radionecrosis were the most common injuries (Table 1), which is consistent with the literature.1,4 The mean treatment time for ORN was 2.2 months, for dermal STRD was 2.1 months, for cystitis was 2.1 months, for proctitis was 2.3 months, and for LRN was 1.8 months.
The difference in the number of HBO sessions among injuries was significant and ranged from 30 to 40 sessions. These results are consistent with HBO protocols, as the number of sessions varies depending on the patients_ individualized needs, but usually, 30 to 40 sessions are applied.1
Chamber pressure of each HBO session varied from 2.0 to 2.5 atmospheres absolute, which is the standard pressure range
Figure 1: PAIN SYMPTOMS POST-HBOT
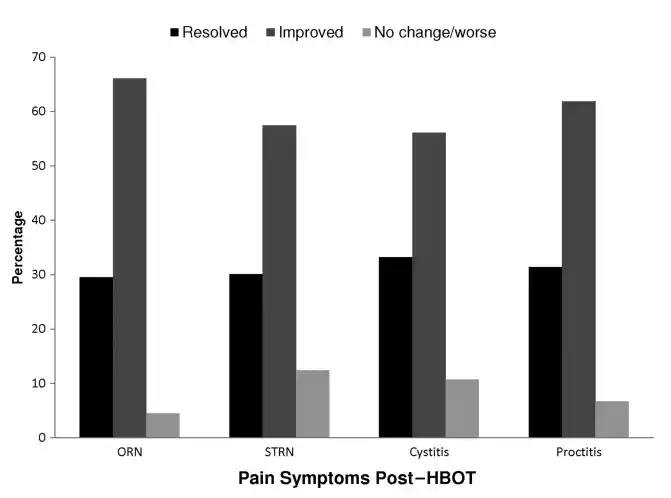
The distribution of postYHBOT outcomes for pain for those patients reporting pain and for which symptom-linked outcomes are available.
Abbreviations: HBOT, hyperbaric oxygen treatment; ORN, osteoradionecrosis; STRN, soft tissue radionecrosis.
applied.1 There was significant variation between types of injury and chamber pressure, with patients with ORN having higher chamber pressures and patients with laryngeal radionecrosis having lower chamber pressures (P < .001). The median time for all groups was 90 minutes, with little variation between groups. The standard duration of an HBO session is 90 to 120 minutes.1
Overall, HBO outcomes were positive with symptoms that improved or resolved to varying from 76.7% to 92.6%, depending on injury type (Table 6). Osteoradionecrosis had the highest mean symptom improvement score (3.24) compared with a mean of 3.04 for LRN (Table 6). Although the mean symptom improvement score between some groups is statistically significant (P values not shown), the differences are probably not clinically meaningful. The most common pre-HBOT symptoms reported in all injury types were pain (Figure 1) and dermal compromise (fistula or wound) (Figures 2 to 5). Hematuria and hematochezia were reported symptoms in all patients with cystitis and proctitis, respectively (Figures 4 and 5). The presenting symptoms of patients in this registry are consistent with the typical clinical presentation of patients with radiation injury.1,27 These findings support the nearly 30 previous studies (most of which were case series) that reported positive HBO outcomes on radionecrosis, cystitis, and proctitis.1,15,16,18-25 The mechanisms by which HBO benefits patients include the promotion of osteogenesis, stimulation of angiogenesis, reduction of inflammatory fibrosis, and increasing tissue vascular density.1,17
Marx et al19 pioneered the first RCT to report the positive benefits of HBO on patients with ORN. There were 74 patients
Figure 2: MAJOR SYMPTOMS OF ORN POST-HBOT
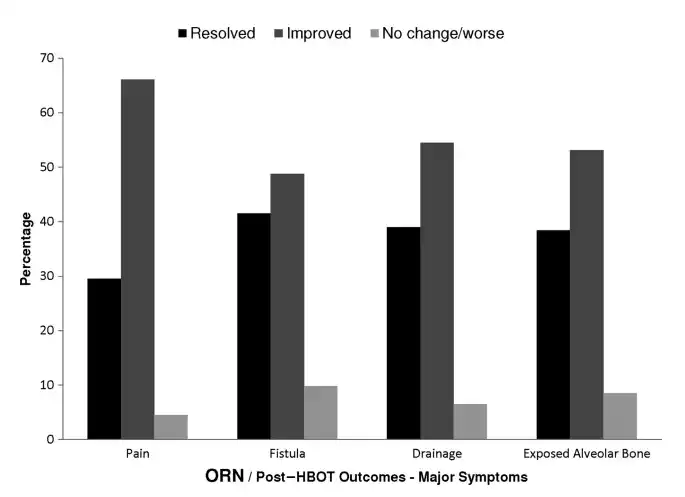
The distribution of postYHBOT outcomes for major symptoms of patients with ORN for which symptom-linked outcomes are available.
Abbreviations: HBOT, hyperbaric oxygen treatment; ORN, osteoradionecrosis.
Figure 3: MAJOR SYMPTOMS OF STRN POST-HBOT
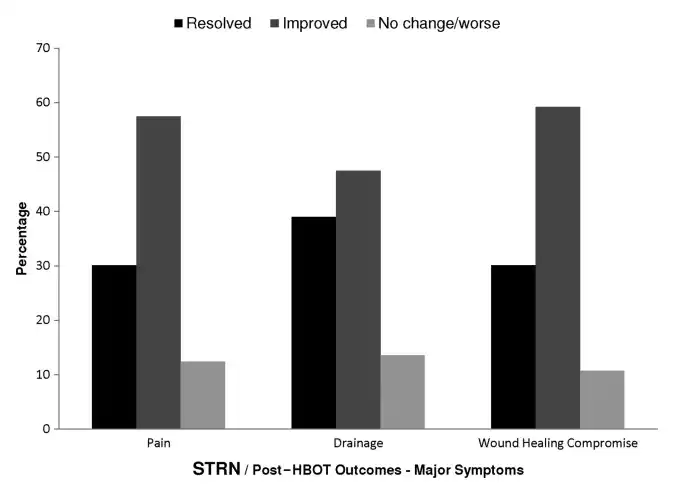
The distribution of post-HBOT outcomes for major symptoms of STRN patients for which symptom-linked outcomes are available.
Abbreviations: HBOT, hyperbaric oxygen treatment; STRN, soft tissue radionecrosis.
from 3 centers included, all of whom underwent dental extraction as a consequence of radiation. Patients were randomized into the penicillin (n = 37, 137 socket wounds) or HBO (n = 37, 156 socket wounds) groups. The HBO group had a significantly less ORN incidence of 5.4% as compared with the 29.9% incidence rate in the penicillin group (P = .005). The authors found that angiogenesis
Figure 4: MAJOR SYMPTOMS OF CYSTITIS POST-HBOT
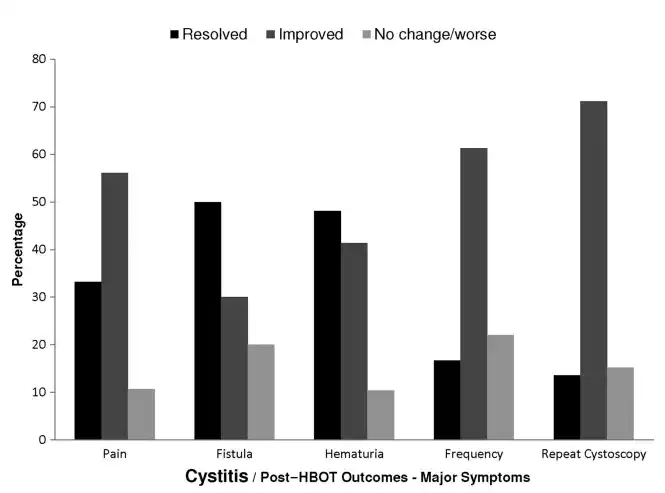
The distribution of postYHBOT outcomes for major symptoms of patients with cystitis for which symptom-linked outcomes are available.
Abbreviation: HBOT, hyperbaric oxygen treatment.
Figure 5: MAJOR SYMPTOMS OF PROCTITIS POST-HBOT
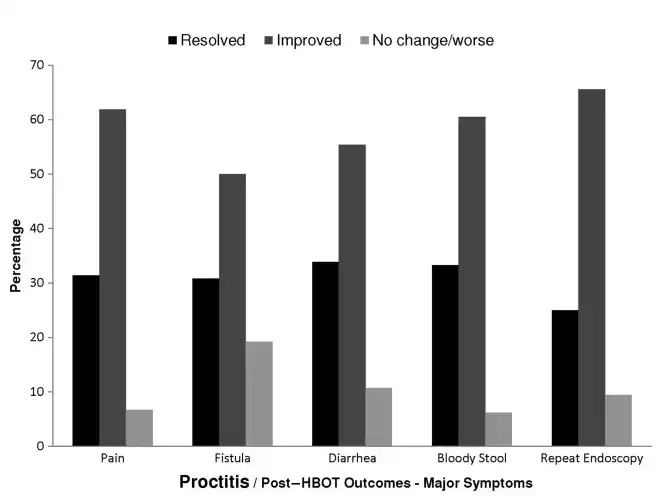
The distribution of post-HBOT outcomes for major symptoms of proctitis patients for which symptom-linked outcomes are available.
Abbreviation: HBOT, hyperbaric oxygen treatment.
was measurable after 8 sessions and improved until 20 sessions, when the levels are then maintained, thus paving the way for HBO protocols.
A more recent double-blind RCT conducted by Freiberger et al20 concluded that HBOT was beneficial to patients with ORN of the jaw. The study had a smaller sample size of 46 patients, who were randomized to the HBO (n = 25) or control (n = 21) groups. Improvement following treatment was observed in 17 HBO patients (68.0%) compared with 8 control patients (38.1%); these differences were significant (P < .05). The mean time for improvement was evaluated, which was not a variable in the current study and significantly different between groups (P < .05). In the HBO group, the mean time was 39.7 weeks (95% confidence interval, 22.4-57.0 weeks) compared with 67.9 weeks (95% confidence interval, 48.4-87.5 weeks) in the control group. The study also evaluated time to healing, unlike the current study, which was also significantly different between groups (P < .05).
There has been 1 placebo-controlled, double-blind RCT conducted with 68 patients that suggests that HBO is not beneficial to ORN.26 Only 19.4% of HBO patients improved at 1 year in comparison with 32.4% in the placebo group. However, this study evaluated HBO as a primary ORN treatment, rather than the recommended adjunctive therapy, and did not follow HBO protocols.1,28 Thus, the results should be considered with caution.
A double-blind crossover RCT evaluated the effectiveness of HBO for radiation proctitis.24 There were 150 patients enrolled; 120 were analyzed. The HBOT significantly improved patient outcomes with an absolute risk reduction of 32% between the HBO and control groups. Approximately 45% of patients, who did not respond to HBOT, had a recurrence, which implicates the need for measured HBO dosing. The authors recommended that no more than 40 sessions be applied. In the current study, patients with proctitis underwent the most sessions (median, 40; or the maximum recommended by the RCT on proctitis) and had the least number of resolved cases (31.3%), but the improvement was still demonstrated.
Evidence is sparser when attempting to compare HBO outcomes with other treatments for radiation injuries. A recent cohort study11 evaluated the outcomes of the Matrix Mandible Preformed Reconstructions Plates ([MMPRP] Synthes CMF, West Chester, Pennsylvania) on 71 patients with ORN. The Matrix Mandible Preformed Reconstructions Plates were placed in 70 patients (98.6%), 10 (14.3%) of which were placed using a transoral approach. The mean operative time was 13.1 minutes. After an average follow-up period of 11.8 months, there were no plate fractures. Nineteen patients (27.1%) had postoperative complications, and 11 (15.7%) required plate removal. Irradiation was administered to 24 patients (34.3%), 62.5% of whom had the majority of complications. Overall, the MMPRP appeared to be a good device to use in reconstructive surgery for ORN with reduced operative time and good outcomes. Improvements in surgical techniques, including microvascular surgery, in recent years, suggest that surgery might ultimately replace HBOT as a better ORN treatment option.29 However, evidence remains low.
Finally, the Cochrane review on the use of HBOT in the treatment of late radiation tissue injury conducted by Bennett et al27 noted evidence that HBOT provided a higher likelihood for mucosal coverage with ORN and statistically significant increased odds of improvement or cure for radiation proctitis and hemimandibulectomy. Furthermore, the authors also concluded that based on the small RCTs already published, for patients with late radiation tissue injury affecting tissues of the head, neck, anus, and rectum, HBOT is associated with improved outcomes.
The main limitations of this study are the fact that it did not have a comparative arm (ie, other treatments) and the level of data characterizing patient comorbidity and symptom severity. A future study could collect additional patient data over a longer time frame to better evaluate post-HBOT treatment so that predictive outcome modeling could be undertaken.
To the authors_ knowledge, the current observational cohort study includes one of the largest radiation injury data sets of HBOT outcomes to date and supports the previous evidence of the effectiveness of HBOT on radiation-induced injuries.
Outcomes from the ACHM RRR, which included 2538 patients with radiation-induced injuries, support the use of HBOT as an adjunctive treatment for radiation injuries. Clinical outcomes following HBOT were positive with symptoms reported as improved or resolved in 76.7% to 92.6% of patients. These findings support the efficacy of the effectiveness of HBOT in radiation-induced injuries. However, stronger evidence in the form of RCTs is still needed to further define the use of HBOT as a standard of care. The ACHM registry effort provides a prototype that can be built on, improved, and, hopefully, replicated to continue to elucidate the clinical efficacy of HBOT.
Jeffrey A. Niezgoda, MD, FACHM, MAPWCA, CHWS, is President, American College of Hyperbaric Medicine; President/Chief Medical Officer, WebCME; President/Chief Medical Officer, AZH; and Industry Consultant for Hyperbaric and Wound Care, Milwaukee, Wisconsin. Thomas E. Serena, MD, FACS, FACHM, MAPWCA, is Founder/Chief Executive Officer/Medical Director, SerenaGroup, Cambridge, Massachusetts; President, American Professional Wound Care Association; and Vice President, American College of Hyperbaric Medicine. Marissa J. Carter, PhD, MAPWCA, is President, Strategic Solutions, Inc, Cody, Wyoming. Drs Niezgoda and Serena have disclosed that the American College of Hyperbaric Medicine has received funding from the Eric P. Kindwall, MD, Foundation. Dr Carter has disclosed that her company is remunerated for participation in review activities such as data monitoring boards, statistical analysis, and end point committees, and received payment for writing or reviewing the manuscript; and she is a consultant to the SerenaGroup. The authors disclose that this study was funded by the American College of Hyperbaric Medicine and by a generous donation from the Eric P. Kindwall, MD, Foundation. Acknowledgments: The authors thank the American College of Hyperbaric Medicine for its financial support of the registry analysis and preparation of this article; the generous donation from the Eric P. Kindwall, MD, Foundation; Ken Kneser and Angela Kujath at the American College of Hyperbaric Medicine for technical design, operation, and maintenance of the registry; Kathy Nelson, RN, and Dawn Knoll, RN, for clinical research support; Kate Baker at WebCME for video production, and education and promotional assistance; and Kristen Eckert (Strategic Solutions) for assistance in writing the manuscript. Submitted December, 30, 2014; accepted in revised form July 28, 2015.